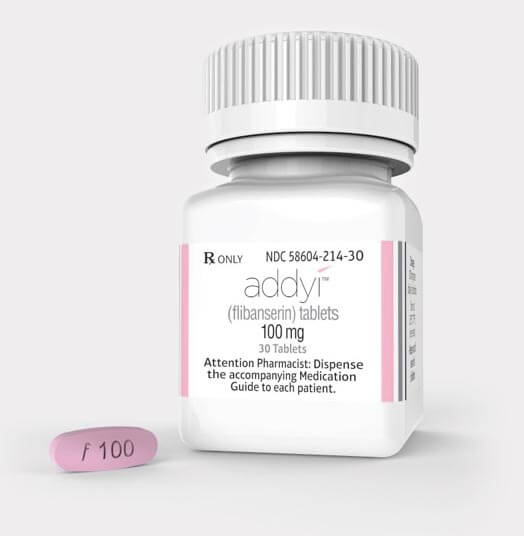
On 18th August 2015 the U.S FDA (Food and Drug Administration) approved the drug Flibanserin, the first and only female treatment for acquired, generalized Hypoactive Sexual Desire Disorder (HSDD).
HSDD affects around 1 in 10 women.
HSDD is acquired when symptoms occur after a period of normal sexual functioning.
Hypoactive Sexual Desire Disorder is a generalized when it occures regardless of the type of sexual activity, sexual partner or the situation.
Flibanserin will only be available in the USA as the brand name Addyi.
There are currently no indications as to when (or if) it will be approved for use in the EU or UK.
 To read the full article, go to: http://www.ukmeds4u.com/news/Drug-information:-Not-legal-in-the-UK/12/Hypoactive-Sexual-Desire-Disorder-(HSDD)-and-Addyi-(Flibanserin)/758/
To read the full article, go to: http://www.ukmeds4u.com/news/Drug-information:-Not-legal-in-the-UK/12/Hypoactive-Sexual-Desire-Disorder-(HSDD)-and-Addyi-(Flibanserin)/758/
For information on sexual health, please go to: www.ukmeds4u.com